Abstract
Mini Review
Bacteriolysis Destruction for Cu- and Zn-Ions Induced Peptidoglycan Cell Wall Based on the Halo-test Results in Cu(NO3)2 and ZnSO4 Solutions against Staphylococcus epidermidis
Tsuneo Ishida* and Reiko Kobayashi
Published: 24 March, 2026 | Volume 9 - Issue 1 | Pages: 010-017
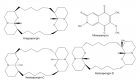
Bacteriolysis of peptidoglycan (PGN) cell wall destruction on cleavage, hydrolysis, and decomposition by copper nitrate and zinc sulfide solutions has been investigated on the ground of the results obtained from halo antibacterial susceptibility tests in metal sulfate solutions against Staphylococcus epidermidis, in which from halo-antibacterial susceptibility tests of metallic ion concentration of 100 mM/L against Staphylococcus epidermidis, the order of bacterial effect for the metal nitrate solutions is as follows, Cu2+>Zn2+ >Ag+>Pb2+>Al3+, and the other, in the metal sulfate solutions, the antibacterial effect order is found to be Zn2+>Cu2+>Ag+>Al3+.
Cu(Ⅱ)-, Zn(Ⅱ)-ions induced bacteriolytic destructions for PGN cell wall on cleavage, hydrolysis, and decomposition within Cu(NO3)2 and ZnSO4 solutions have been elucidated that Cu(Ⅱ) and Zn(Ⅱ) ions can inhibit PGN biosynthesis Transpeptidase (TP)/Trans-glycosylase (TG) and PGN elongation, PGN cleavage occurs by copper ions- and zinc ions-containing autolysins amidase; AmiE, Rv3717, AmiA, PGN cell wall is hydrolyzed by copper-, and zinc-containing enzymes; PGRPs, MurA, LytA, LytM=Endopeptidase, Hydrolase LytB, and PGN decomposition by copper-, zinc-containing PGN inhibitive elongation occurs. Thus, Cu(Ⅱ) and Zn(Ⅱ) within Cu(NO3)2 and ZnSO4 solutions inhibit PGN biosynthesis and activate bacterial PGN autolysins against Staphylococcus epidermidis.
Read Full Article HTML DOI: 10.29328/journal.ijcmbt.1001034 Cite this Article Read Full Article PDF
Keywords:
Zinc(Ⅱ) and copper(Ⅱ); Halo-test; Copper nitrate and zinc sulfate; PGN biosynthesis; Autolysin and elongation; S. aureus PGN cleavage; Hydrolysis and decomposition
References
- Toplitsch D, Lackner JM, Schwan AM, Hinterer A, Stögmüller P, Horn K, et al. Antimicrobial activity of a novel Cu(NO3)2-containing sol–gel surface under different testing conditions. Materials. 2021;14:6488:1-14. Available from: https://doi.org/10.3390/ma14216488
- Abdalkader FA. Antibacterial effect of different concentrations of zinc sulfate on multidrug resistant pathogenic bacteria. Sys Rev Pharm. 2020;11(3):282-288. Available from: http://dx.doi.org/10.5530/srp.2019.2.04
- Murphey ED, Sherwood ER. Pretreatment with the gram-positive bacterial cell wall molecule peptidoglycan improves bacterial clearance and decreases inflammation and mortality in mice challenged with Pseudomonas aeruginosa. Microbes Infect. 2008;10(12-13):1244-1250. Available from: https://doi.org/10.1016/j.micinf.2008.07.021
- Ishida T. Insights into metallic Ag+, Cu2+, Zn2+ ions-induced bacteriolytic mechanism against S. aureus and E. coli. Catal Res. 2023;3(1):1-12. Available from: https://www.jcatalysis.com/articles/insights-into-metallic-ag-cu2-zn2-ions-induced-bacteriolytic-mechanism-against-s-aureus-and-e-coli.pdf
- Büttner FM, Zoll S, Nega M, Götz F, Stehle T. Structure-function analysis of Staphylococcus aureus amidase reveals the determinants of peptidoglycan recognition and cleavage. J Biol Chem. 2014;289(16):11083-11094. Available from: https://doi.org/10.1074/jbc.m114.557306
- Guan R, Roychowdhury A, Ember B, Kumar S, Boons GJ, Mariuzza RA. Structural basis for peptidoglycan binding by peptidoglycan recognition proteins. Proc Natl Acad Sci U S A. 2004;101(49):17168-17173. Available from: https://doi.org/10.1073/pnas.0407856101
- Liu X, Grabherr HM, Willmann R, Kolb D, Brunner F, Bertsche U, Kühner D, Franz-Wachtel M, Amin B, Felix G, Ongena M, Nürnberger T, Gust AA. Host-induced bacterial cell wall decomposition mediates pattern-triggered immunity in Arabidopsis. Elife. 2014;3:1-24. Available from: https://doi.org/10.7554/elife.01990
- Ishida T. Bacteriolyses of bacterial cell walls by Cu(II) and Zn(II) ions based on antibacterial results of dilution medium method and halo antibacterial test. J Adv Res Biotechnol. 2017;2(2):1-12. Available from: https://symbiosisonlinepublishing.com/biotechnology/biotechnology20.pdf
- Ishida T, Kobayashi R, Inose W. Anti-bacterial activity on halo antibacterial susceptibility test by metal sulfates and nitrides solutions. J Japan Res Inst Adv Copper-Base Mater Technol. 2017;56(1):329-333.
- Crichton RT, Shioya M. Biological inorganic chemistry. Tokyo: Tokyo Kagaku-Dojin Limited; 2016;175-188. Available from: https://miguelprudencio.com/wp-content/uploads/Crichton-Biological-Inorganic-Chemistry-An-Introduction.pdf
- Zoll S, Patzold B, Schlag M, Gotz F, Kalbacher H, Stehle T. Structural basis of cell wall cleavage by a staphylococcal autolysin. PLoS Pathog. 2010;6(3):e1000807. Available from: https://doi.org/10.1371/journal.ppat.1000807
- Humann J, Lenz LL. Bacterial peptidoglycan-degrading enzymes and their impact on host muropeptidase detection. J Innate Immun. 2009;1(2):88-97. Available from: https://doi.org/10.1159/000181181
- Ishida T. Anti-bacterial mechanism for metallic Ag+, Cu2+, Zn2+ ions-induced bacteriolysis on disruptive OM Lpp and PGN inhibitive elongations against S. aureus and E. coli. Mathews J Cytol Histol. 2022;6(1):1-13. Available from: https://www.mathewsopenaccess.com/scholarly-articles/anti-bacterial-mechanism-for-metallic-ag-cu2-zn2-ions-induced-bactertiolysis-on-disruptive-om-lpp-and-pgn-inhibitive-elongations-against-s-aureus-and-e-coli.pdf
- Ishida T. Antibacterial mechanism of bacteriolyses of bacterial cell walls by zinc(II) ion induced activations of PGN autolysins, and DNA damages. J Genes Proteins. 2017;1(1):1-7. Available from: https://www.scitechnol.com/peer-review-pdfs/antibacterial-mechanism-of-bacteriolyses-of-bacterial-cell-walls-by-zinc-ion-induced-activations-of-pgn-autolysins-and-dna-damages-juTB.pdf
- Wang M, Liu LH, Wang S, Li X, Lu X, Gupta D, et al. Human peptidoglycan recognition proteins require zinc to kill both gram-positive and gram-negative bacteria and are synergistic with antibacterial peptides. J Immunol. 2007;178:3116-3125. Available from: https://doi.org/10.4049/jimmunol.178.5.3116
- Conrady DG, Brescia CC, Horii K, Weiss AA, Hassett DJ, Herr AB. A zinc-dependent adhesion module is responsible for intercellular adhesion in staphylococcal biofilms. Proc Natl Acad Sci U S A. 2008;105(49):19456-19461. Available from: https://doi.org/10.1073/pnas.0807717105
- Atilano ML, Pereira PM, Yates J, Reed P, Veiga H, Pinho MG, et al. Teichoic acids are temporal and spatial regulators of peptidoglycan cross-linking in Staphylococcus aureus. Proc Natl Acad Sci U S A. 2010;107(44):18991-18996. Available from: https://doi.org/10.1073/pnas.1004304107
- Ong CY, Walker MJ, McEwan AG. Zinc disrupts central carbon metabolism and capsule biosynthesis in Streptococcus pyogenes. Sci Rep. 2015;5:10799. Available from: https://www.nature.com/articles/srep10799
- Chandrangsu P, Helmann JD. Intracellular Zn(II) intoxication leads to dysregulation of the PerR regulon resulting in heme toxicity in Bacillus subtilis. PLoS Genet. 2016;12(12):e1006515. Available from: https://doi.org/10.1371/journal.pgen.1006515
- Brazel EB, Tan A, Neville SL, Iverson AR, Udagedara SR, Cunningham BA, et al. Dysregulation of Streptococcus pneumoniae zinc homeostasis breaks ampicillin resistance in a pneumonia infection model. Cell Rep. 2022;38(2):110202. Available from: https://doi.org/10.1016/j.celrep.2021.110202
- Ishida T. Mechanism of antibacterial activities of Cu(II) ions against Staphylococcus aureus and Escherichia coli on the ground of results obtained from dilution medium method. Virol Immunol J. 2017;1(3):1-8. Available from: https://medwinpublishers.com/VIJ/VIJ16000117.pdf
- Mahmood WS. Controlling of bacterial elongation growth by studying the factors affecting it. World J Mod Res Methodol. 2023;2(11):2835-3072. Available from: https://doi.org/10.15226/2475-4714/2/2/00120
- Prigozhin DM, Mavrici D, Huizar JP, Vansell HJ, Alber T. Structural and biochemical analyses of Mycobacterium tuberculosis N-acetylmuramyl-L-alanine amidase Rv3717 point to a role in peptidoglycan fragment recycling. J Biol Chem. 2013;288(44):31549-31555. Available from: https://doi.org/10.1074/jbc.M113.510792
- Zoll S, Pätzold B, Schlag M, Götz F, Kalbacher H, Stehle T. Structural basis of cell wall cleavage by a staphylococcal autolysin. PLoS Pathog. 2010;6:1-13. Available from: https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1000807
- Argüello JM, Raimunda D, Padilla-Benavides T. Mechanisms of copper homeostasis in bacteria. Front Cell Infect Microbiol. 2013;3:73. Available from: https://doi.org/10.3389/fcimb.2013.00073
- Guan R, Roychowdury A, Ember B, Kumar S, Boons GJ, Mariuzza RA. Crystal structure of a peptidoglycan recognition protein in complex with a muramyl tripeptide from gram-positive bacteria. J Endotoxin Res. 2005;11(1):1-6. Available from: https://doi.org/10.1179/096805105225006713
- Carroll SA, Hain T, Technow U, Darji A, Pashalidis P, Joseph SW, et al. Identification and characterization of a peptidoglycan hydrolase, MurA of Listeria monocytogenes, a muramidase needed for cell separation. J Bacteriol. 2003;185(23):6801-6808. Available from: https://doi.org/10.1128/jb.185.23.6801-6808.2003
- Paton JC, Andrew PW, Boulnois GJ, Mitchell TJ. Molecular analysis of the pathogenicity of Streptococcus pneumoniae: the role of pneumococcal proteins. Annu Rev Microbiol. 1993;47:89-115. Available from: https://doi.org/10.1146/annurev.mi.47.100193.000513
- Mellroth P, Sandalova T, Kikhney A, Vilaplana F, Hesek D, Lee M, et al. Structural and functional insights into peptidoglycan access for the lytic amidase LytA of Streptococcus pneumoniae. mBio. 2014;5(1):1-10. Available from: https://doi.org/10.1128/mbio.01120-13
- Jagielska E, Chojnacka O, Sabata I. LytM fusion with SH3b-like domain expands its activity to physiological conditions. Microb Drug Resist. 2016;22(6):461-469. Available from: https://doi.org/10.1089/mdr.2016.0053
- Bai XH, Chen HJ, Jiang YL, Wen Z, Huang Y, Cheng W, Li Q, Qi L, Zhang JR, Chen Y, Zhou CZ. Structure of pneumococcal peptidoglycan hydrolase LytB reveals insights into bacterial cell wall remodeling and pathogenesis. J Biol Chem. 2014;289:1-10. Available from: https://doi.org/10.1074/jbc.m114.579714
- Godoy-Gallardo M, Hoyos-Nogué M, Eckhard U, Gil FJ, Delgado LM, Perez RA, de Roo Puente YJD. Antibacterial approaches in tissue engineering using metal ions and nanoparticle. Bioact Mater. 2021. Available from: https://doi.org/10.1016/j.bioactmat.2021.04.033
- Božić Cvijan B, Korac Jaći J, Bajceti M. The impact of copper ions on the activity of antibiotic drugs. Molecules. 2023;28:5133:1-15. Available from: https://doi.org/10.3390/molecules28135133
- Wu W, Wang X, Zhang W, Reed W, Samet JM, Whang YE, Ghio AJ. Zinc-induced PTEN protein degradation through the proteasome pathway in human airway epithelial cells. J Biol Chem. 2003;278(30):28258-28263. Available from: https://doi.org/10.1074/jbc.m303318200
- Ishida T. Antibacterial mechanism of bacteriolyses of bacterial cell walls by zinc(II) ion induced activations of PGN autolysins, and DNA damages. J Genes Proteins. 2017;1(1):1-7. Available from: https://www.scitechnol.com/peer-review-pdfs/antibacterial-mechanism-of-bacteriolyses-of-bacterial-cell-walls-by-zinc-ion-induced-activations-of-pgn-autolysins-and-dna-damages-juTB.pdf
- Eron SJ, MacPherson DJ, Dagbay KB, Hardy JA. Multiple mechanisms of zinc-mediated inhibition for the apoptotic caspases -3, -6, -7, and -8. ACS Chem Biol. 2018;13(5):1279-1290. Available from: https://doi.org/10.1021/acschembio.8b00064
Figures:
Similar Articles
-
Bacteriolysis Destruction for Cu- and Zn-Ions Induced Peptidoglycan Cell Wall Based on the Halo-test Results in Cu(NO3)2 and ZnSO4 Solutions against Staphylococcus epidermidisTsuneo Ishida*,Reiko Kobayashi. Bacteriolysis Destruction for Cu- and Zn-Ions Induced Peptidoglycan Cell Wall Based on the Halo-test Results in Cu(NO3)2 and ZnSO4 Solutions against Staphylococcus epidermidis. . 2026 doi: 10.29328/journal.ijcmbt.1001034; 9: 010-017
Recently Viewed
-
Morning and Evening Exercise with or without High-Pressure Intervals have Different Effects on Controlling Blood Sugar and Glucose Fluctuations in People with Type 1 and Type 2 DiabetesReza Sheikh*. Morning and Evening Exercise with or without High-Pressure Intervals have Different Effects on Controlling Blood Sugar and Glucose Fluctuations in People with Type 1 and Type 2 Diabetes . Int J Bone Marrow Res. 2023: doi: 10.29328/journal.ijbmr.1001016; 6: 001-003
-
B-ultrasound-guided Intrahepatic Infusion of Autologous Bone Marrow Cells for Decompensated CirrhosisBaochi Liu*, Xiong Gao, Yuanhuai Chen, Qiqiang Dong, Jingbo Wang, Baisong Zhao. B-ultrasound-guided Intrahepatic Infusion of Autologous Bone Marrow Cells for Decompensated Cirrhosis. Int J Bone Marrow Res. 2024: doi: 10.29328/journal.jbmr.1001017; 7: 001-006
-
Impact of Intravenous Busulfan Pharmacokinetics on Safety in Pediatric Patients who have undergone Hematopoietic Stem Cell TransplantOmar AL Mofleh*,Noha Awadalla,Amal AL Shafi,Lina Husain,Hanan AL Musabeh,Saad AL Daama. Impact of Intravenous Busulfan Pharmacokinetics on Safety in Pediatric Patients who have undergone Hematopoietic Stem Cell Transplant. Int J Bone Marrow Res. 2024: doi: 10.29328/journal.ijbmr.1001018; 7: 007-012
-
Exploring the Potential of Medicinal Plants in Bone Marrow Regeneration and Hematopoietic Stem Cell TherapyUgwu Okechukwu Paul-Chima*,Alum Esther Ugo. Exploring the Potential of Medicinal Plants in Bone Marrow Regeneration and Hematopoietic Stem Cell Therapy. Int J Bone Marrow Res. 2025: doi: 10.29328/journal.ijbmr.1001019; 8: 001-005
-
Deep Learning-Powered Genetic Insights for Elite Swimming Performance: Integrating DNA Markers, Physiological Biometrics and Performance AnalyticsRahul Kathuria,Reeta Devi,Asadi Srinivasulu*. Deep Learning-Powered Genetic Insights for Elite Swimming Performance: Integrating DNA Markers, Physiological Biometrics and Performance Analytics. Int J Bone Marrow Res. 2025: doi: 10.29328/journal.ijbmr.1001020; 8: 006-015
Most Viewed
-
Physical Performance in the Overweight/Obesity Children Evaluation and RehabilitationCristina Popescu, Mircea-Sebastian Șerbănescu, Gigi Calin*, Magdalena Rodica Trăistaru. Physical Performance in the Overweight/Obesity Children Evaluation and Rehabilitation. Ann Clin Endocrinol Metabol. 2024 doi: 10.29328/journal.acem.1001030; 8: 004-012
-
Hypercalcaemic Crisis Associated with Hyperthyroidism: A Rare and Challenging PresentationKarthik Baburaj*, Priya Thottiyil Nair, Abeed Hussain, Vimal MV. Hypercalcaemic Crisis Associated with Hyperthyroidism: A Rare and Challenging Presentation. Ann Clin Endocrinol Metabol. 2024 doi: 10.29328/journal.acem.1001029; 8: 001-003
-
Effects of dietary supplementation on progression to type 2 diabetes in subjects with prediabetes: a single center randomized double-blind placebo-controlled trialSathit Niramitmahapanya*,Preeyapat Chattieng,Tiersidh Nasomphan,Korbtham Sathirakul. Effects of dietary supplementation on progression to type 2 diabetes in subjects with prediabetes: a single center randomized double-blind placebo-controlled trial. Ann Clin Endocrinol Metabol. 2023 doi: 10.29328/journal.acem.1001026; 7: 00-007
-
Exceptional cancer responders: A zone-to-goDaniel Gandia,Cecilia Suárez*. Exceptional cancer responders: A zone-to-go. Arch Cancer Sci Ther. 2023 doi: 10.29328/journal.acst.1001033; 7: 001-002
-
Ectopic adrenal tissue at the spermatic cordAbdallah Chaachou,Nizar Cherni,Wael Ferjaoui*,Mohamed Dridi,Samir Ghozzi. Ectopic adrenal tissue at the spermatic cord. J Clin Med Exp Images. 2022 doi: 10.29328/journal.jcmei.1001024; 6: 001-002

If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."